 
Step 3: As the hydrocarbon is neutral so, the total charge z is 0 and the product zme is also 0. Apply the formula for molar mass calculation. Step 2: Now, we know the formula of the hydrocarbon. 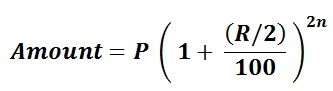
suppose the estimated formula is C18H34O2 (oleic acid). Use the spectroscopy technique to determine the number of carbon and hydrogen present in the sample. The amount of CO2 and H2O released tells the number of carbon, hydrogen, and oxygen present. Step 1: As we know that hydrocarbons consist of carbon and hydrogen, so we do the combustion reaction of the sample in the presence of oxygen. The answer is the molar mass of the sample compound.Įxample Calculate the molar mass of the hydrocarbon present in the given sample.Add all the NiMi values and subtract from the zme value.Multiply the charge number (z) of the sample compound with the electron mass (me).Multiply the atomic mass of each atom with its total number (NiMi).Calculate the number of each atom present in the sample compound (Ni).Where Ni is the number of each atom, Mi is the atomic mass of each atom, z is the total charge number of the unknown compound, and me is the electron mass. The molar mass of the sample compound can be calculated using the given formula: Equilibrium constants and rate of reaction.įollowing are some physical techniques used in the elemental analysis of an unknown sample:.Chemical names of the reactants and the product.Chemical structure of the reactants and the product.In the case of an unknown compound, it would determine the:įor an unbalanced chemical equation, it would determine the: Benefits of the Elemental Composition Calculator The calculator would predict the balanced reaction equation and estimated products also. For an unknown reaction equation, enter the unbalanced equation or only enter the reactants. If you don’t know the formula of the compound then you may enter the name of the compound (e.g., stearic acid). You can enter the formula (e.g., C4H4O) of the compound to get the information about it. The qualitative analysis tells the type of atoms, and elements present in the sample while quantitative analysis tells the number of atoms, and elements present in the sample. The analysis may be qualitative and quantitative. 
The analysis includes the determination of empirical formula, molecular formula, and molar mass of the sample compound by analyzing the elements present in it. It determines the type and number of atoms, elements, and ions present in a sample. It is the analysis of an unknown sample (like water, air, soil, etc.). This article gives information about the elemental analysis process and how you can get quick results using this calculator. This online calculator gives complete information about the given compound. These analysis calculations are lengthy and time-consuming but our calculator has made them quick and easy to understand. This tool is used to calculate the molar mass of a compound during the elemental analysis. If you want to ionic formula of a compound, them have a look at Chemistrycalc.Com and hit the Calculator Ionic Formula link to get the accurate answer without fail.Introduction to Elemental Analysis Calculator The ion name is sodium cation or sodium ion Question: Calculate the net ionic charge of Na + The ionic formula can be written in 2 ways, one is general method and the other is a criss-cross method. The net ionic charge is nothing but the number of cations or anions a chemical compound is having. In an ionic compound, the number of cations and anions are equal. The positively charged ions are called cations and negatively charged ions are called anions. The formed compound is neutral as it has positively charged ions and negatively charged ions. An ionic compound is defined as a chemical compound that is made up of ions that are held together by the electrostatic force known as ionic bonding. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
